COVID-19
COVID-19 (SARS-CoV-2) Detection and Research
PCR Biosystems is committed to providing researchers, developers and diagnostic kit manufacturers with the PCR products and services they need in the global response to COVID-19.
We understand there are many challenges to be faced during this crisis, not least testing for the SARS-CoV-2 virus on a vast scale. To meet the needs of our customers we have massively increased manufacture of critical reagents for rapid and sensitive RT-qPCR. You can read more about our scaled-up operations here.
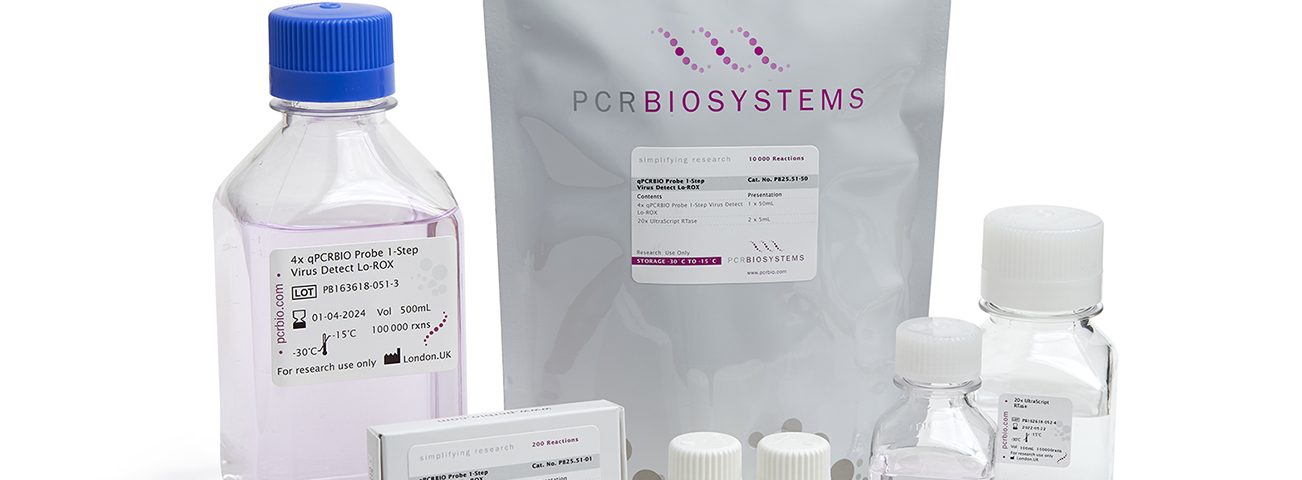
To support high throughput diagnostic workflows, we’ve introduced 50mL and 500mL bulk sizes of our RT-qPCR mixes into to our standard portfolio to provide greater consistency, convenience and value in testing for COVID-19.
We manufacture under a rigorous ISO 9001 and ISO 13485 quality system meaning our products and processes meet (and exceed) the required standards and regulations for the manufacturing of medical device components.
With over 180 years of PCR experience within our team, our experts are on hand to help optimise assay conditions and increase testing turnaround times specific to your set up. Please contact us to find out more.
We will continue to update this page with new information on how our products and services can support your COVID-19 research and diagnostic development as the outbreak evolves. For up to date information on the national and international response, visit the WHO, CDC, and PHE websites.

